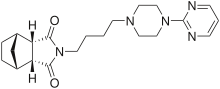
Tandospiron
(IUPAC ) ime
(1R ,2R ,6S ,7S )-44-[4-(pirimidin-2-il)piperazin-1-il]butil4-azatriciklo[5.2.1.02,6 ]dekan-3,5-dion
Klinički podaci
AHFS/Drugs.com
Internacionalno ime leka
Identifikatori
CAS broj
112457-95-1
ATC kod
nije dodeljen
PubChem [1] [2] 91273
ChemSpider [3] 82421
UNII
190230I669 Y
ChEMBL [4] CHEMBL274047 Y
Hemijski podaci
Formula
C 21 H 29 N 5 O 2
Mol. masa
383,487 g/mol
SMILES
eMolekuli PubHem
InChI InChI=1S/C21H29N5O2/c27-19-17-15-4-5-16(14-15)18(17)20(28)26(19)9-2-1-8-24-10-12-25(13-11-24)21-22-6-3-7-23-21/h3,6-7,15-18H,1-2,4-5,8-14H2/t15-,16+,17+,18- Y Y
Farmakokinetički podaci
Poluvreme eliminacije
1,2–1,4 sata
Farmakoinformacioni podaci
Trudnoća
?
Pravni status
℞ Prescription only
Način primene
Oralno
Tandospiron (Sediel , metanopiron ) je anksiolitik i antidepresiv koji se koristi u Kini i Japanu . On je član azapironske i piperazinske hemijske klase, i blisko je srodan sa drugim agensima poput buspirona i gepirona .
Tandospiron deluje kao potentan i selektivan parcijalni agonist 5-HT1A receptora , sa Ki afinitetom od 27 ± 5 nM[5] [6] [7] 5-HT2A (1,300 ± 200), 5-HT2C (2,600 ± 60), α1 -adrenergički (1,600 ± 80), α2 -adrenergički (1,900 ± 400), D1 (41,000 ± 10,000), i D2 (1,700 ± 300) receptore , i esencijalno je neaktivan na 5-HT1B , 5-HT1D , β-adrenergičkom , i muskarinskim acetilholinskim receptorima , serotoniskom transportertu (SERT), i benzodiazepinskom (BDZ) alosternom mestu GABAA receptora (svi od kojih su > 100.000).[5] α2 -adrenergičkom receptoru putem njegovog aktivnog metabolita 1-(2-pirimidinil)piperazin (1-PP).[8] [9]
Tandospiron se može sintetisati na sledeće način:[10]
↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.” . Drug Discov Today 15 (23-24): 1052-7. DOI :10.1016/j.drudis.2010.10.003 . PMID 20970519 . edit ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4 : 217-241. DOI :10.1016/S1574-1400(08)00012-1 . ↑ Hettne KM, Williams AJ, van Mulligen EM, Kleinjans J, Tkachenko V, Kors JA. (2010). „Automatic vs. manual curation of a multi-source chemical dictionary: the impact on text mining” . J Cheminform 2 (1): 3. DOI :10.1186/1758-2946-2-3 . PMID 20331846 . edit ↑ Gaulton A, Bellis LJ, Bento AP, Chambers J, Davies M, Hersey A, Light Y, McGlinchey S, Michalovich D, Al-Lazikani B, Overington JP. (2012). „ChEMBL: a large-scale bioactivity database for drug discovery”. Nucleic Acids Res 40 (Database issue): D1100-7. DOI :10.1093/nar/gkr777 . PMID 21948594 . edit ↑ 5,0 5,1 Hamik; Oksenberg, D; Fischette, C; Peroutka, SJ (1990). „Analysis of tandospirone (SM-3997) interactions with neurotransmitter receptor binding sites”. Biological Psychiatry 28 (2): 99–109. DOI :10.1016/0006-3223(90)90627-E . PMID 1974152 . ↑ Tanaka; Tatsuno, T; Shimizu, H; Hirose, A; Kumasaka, Y; Nakamura, M (1995). „Effects of tandospirone on second messenger systems and neurotransmitter release in the rat brain”. General pharmacology 26 (8): 1765–72. DOI :10.1016/0306-3623(95)00077-1 . PMID 8745167 . ↑ Yabuuchi, Kazuki; Tagashira, Rie; Ohno, Yukihiro (2004). „Effects of tandospirone, a novel anxiolytic agent, on human 5-HT1A receptors expressed in Chinese hamster ovary cells (CHO cells)”. Biogenic Amines 18 (3): 319. DOI :10.1163/1569391041501933 . ↑ Blier; Curet, O; Chaput, Y; De Montigny, C (1991). „Tandospirone and its metabolite, 1-(2-pyrimidinyl)-piperazine--II. Effects of acute administration of 1-PP and long-term administration of tandospirone on noradrenergic neurotransmission” . Neuropharmacology 30 (7): 691–701. DOI :10.1016/0028-3908(91)90176-C . PMID 1681447 . ↑ Miller; Thompson, ML; Byrnes, JJ; Greenblatt, DJ; Shemer, A (1992). „Kinetics, brain uptake, and receptor binding of tandospirone and its metabolite 1-(2-pyrimidinyl)-piperazine”. Journal of Clinical Psychopharmacology 12 (5): 341–5. PMID 1362206 . ↑ Yevich, Joseph P.; New, James S.; Smith, David W.; Lobeck, Walter G.; Catt, John D.; Minielli, Joseph L.; Eison, Michael S.; Taylor, Duncan P. i dr.. (1986). „Synthesis and biological evaluation of 1-(1,2-benzisothiazol-3-yl)- and (1,2-benzisoxazol-3-yl)piperazine derivatives as potential antipsychotic agents”. Journal of Medicinal Chemistry 29 (3): 359–69. DOI :10.1021/jm00153a010 . PMID 2869146 .
Serotoninski modulatori i stimulatori (SMS)
Neselektivni
MAOA -Selektivni
MAOB -Selektivni
Agonisti :
5-FNE •
6-FNE •
Amidefrin •
Anisodamin •
Anisodin •
Cirazolin •
Dipivefrin •
Dopamin •
Efedrin •
Epinefrin (Adrenalin) •
Etilefrin •
Etilnorepinefrin •
Indanidin •
Levonordefrin •
Metaraminol •
Metoksamin •
Metildopa •
Midodrin •
Nafazolin •
Norepinefrin (Noradrenalin) •
Oktopamin •
Oksimetazolin •
Fenilefrin •
Fenilpropanolamin •
Pseudoefedrin •
Sinefrin •
Tetrahidrozolin Antagonisti :
Abanohil •
Adimolol •
Ajmalicin •
Alfuzosin •
Amosulalol •
Arotinolol •
Atiprosin •
Benoksatian •
Buflomedil •
Bunazosin •
Karvedilol •
CI-926 •
Korinantin •
Dapiprazol •
DL-017 •
Domesticin •
Doksazosin •
Eugenodilol •
Fenspirid •
GYKI-12,743 •
GYKI-16,084 •
Indoramin •
Ketanserin •
L-765,314 •
Labetalol •
Mefendioksan •
Metazosin •
Monatepil •
Moksisilit (Timoksamin) •
Naftopidil •
Nantenin •
Neldazosin •
Nicergolin •
Niguldipin •
Pelanserin •
Fendioksan •
Fenoksibenzamin •
Fentolamin •
Piperoksan •
Prazosin •
Hinazosin •
Ritanserin •
RS-97,078 •
SGB-1,534 •
Silodosin •
SL-89.0591 •
Spiperon •
Talipeksol •
Tamsulosin •
Terazosin •
Tibalosin •
Tiodazosin •
Tipentosin •
Tolazolin •
Trimazosin •
Upidosin •
Urapidil •
Zolertin * Mnogi TCA , TeCA , antipsihotici , ergolini , i neki piperazini kao što su buspiron , trazodon , nefazodon , etoperidon , i mepiprazol takođe antagoniziraju α1 -adrenergičke receptore, što doprinosi njihovim nuspojavama.
Agonisti :
Azapironi :
Alnespiron •
Binospiron •
Buspiron •
Enilospiron •
Eptapiron •
Gepiron •
Ipsapiron •
Perospiron •
Revospiron •
Tandospiron •
Tiospiron •
Umespiron •
Zalospiron ;
Antidepresivi :
Etoperidon •
Nefazodon •
Trazodon ;
Antipsihotici :
Aripiprazol •
Asenapin •
Klozapin •
Kvetiapin •
Ziprasidon ;
Ergolini :
Dihidroergotamin •
Ergotamin •
Lisurid •
Metisergid •
LSD ;
Triptamini :
5-CT •
5-MeO-DMT •
5-MT •
Bufotenin •
DMT •
Indorenat •
Psilocin •
Psilocibin ;
Drugi :
8-OH-DPAT •
Adatanserin •
Bay R 1531 •
Befiradol •
BMY-14802 •
Kanabidiol •
Dimemebfe •
Ebalzotan •
Eltoprazin •
F-11,461 •
F-12,826 •
F-13,714 •
F-14,679 •
F-15,063 •
F-15,599 •
Flesinoksan •
Flibanserin •
Lesopitron •
Lu AA21004 •
LY-293,284 •
LY-301,317 •
MKC-242 •
NBUMP •
Osemozotan •
Oksaflozan •
Pardoprunoks •
Piklozotan •
Rauvolscin •
Repinotan •
Roksindol •
RU-24,969 •
S 14,506 •
S-14,671 •
S-15,535 •
Sarizotan •
SSR-181,507 •
Sunepitron •
U-92,016-A •
Urapidil •
Vilazodon •
Ksaliproden •
Johimbin Antagonisti :
Antipsihotici :
Iloperidon •
Risperidon •
Sertindol ;
Beta blokatori :
Alprenolol •
Cianopindolol •
Jodocianopindolol •
Oksprenolol •
Pindobind •
Pindolol •
Propranolol •
Tertatolol ;
Drugi :
AV965 •
BMY-7,378 •
CSP-2503 •
Dotarizin •
Flopropion •
GR-46611 •
Isamoltan •
Lekozotan •
Metitepin/Metiotepin •
MPPF •
NAN-190 •
PRX-00023 •
Robalzotan •
S-15535 •
SB-649,915 •
SDZ 216-525 •
Spiperon •
Spiramid •
Spiroksatrin •
UH-301 •
WAY-100,135 •
WAY-100,635 •
Ksilamidin Agonisti :
Lisergamidi :
Dihidroergotamin •
Metisergid ;
Triptani :
Almotriptan •
Avitriptan •
Eletriptan •
Frovatriptan •
Naratriptan •
Rizatriptan •
Sumatriptan •
Zolmitriptan ;
Triptamini :
5-CT •
5-Etil-DMT •
5-MT •
5-(Noniloksi)triptamin ;
Drugi :
CP-135,807 •
CP-286,601 •
GR-46611 •
L-694,247 •
L-772,405 •
PNU-109,291 •
PNU-142,633 Antagonisti :
Lisergamidi :
Metergolin ;
Drugi :
Alniditan •
BRL-15,572 •
Elzasonan •
GR-127,935 •
Ketanserin •
LY-310,762 •
LY-367,642 •
LY-456,219 •
LY-456,220 •
Metitepin/Metiotepin •
Ritanserin •
Johimbin •
Ziprasidon
Agonisti :
Fenetilamini :
2C-B •
2C-E •
2C-I •
2C-T-2 •
2C-T-7 •
2C-T-21 •
DOB •
DOC •
DOI •
DOM •
MDA •
MDMA •
Meskalin ;
Piperazini :
Aripiprazol •
mCPP •
TFMPP ;
Triptamini :
5-CT •
5-MeO-α-ET •
5-MeO-α-MT •
5-MeO-DET •
5-MeO-DiPT •
5-MeO-DMT •
5-MeO-DPT •
5-MT •
α-ET •
α-Metil-5-HT •
α-MT •
Bufotenin •
DET •
DiPT •
DMT •
DPT •
Psilocin •
Psilocibin ;
Drugi :
A-372,159 •
AL-38022A •
CP-809,101 •
Dimemebfe •
Lorkaserin •
Medifoksamin •
MK-212 •
Org 12,962 •
ORG-37,684 •
Oksaflozan •
PNU-22394 •
Ro60-0175 •
Ro60-0213 •
Vabicaserin •
WAY-629 •
WAY-161,503 •
YM-348 Antagonisti :
Atipični antipsihotici :
Klozapin •
Iloperidon •
Melperon •
Olanzapin •
Paliperidon •
Pimozid •
Hetiapin •
Risperidon •
Sertindol •
Ziprasidon •
Zotepin ;
Tipični antipsihotici :
Hlorpromazin •
Loksapin •
Pipamperon ;
Antidepresivi :
Agomelatin •
Amitriptilin •
Amoksapin •
Aptazapin •
Etoperidon •
Fluoksetin •
Mianserin •
Mirtazapin •
Nefazodon •
Nortriptilin •
Trazodon ;
Drugi :
Adatanserin •
Cinanserin •
Ciproheptadin •
Deramciklan •
Dotarizin •
Eltoprazin •
Esmirtazapin •
FR-260,010 •
Ketanserin •
Ketotifen •
Latrepirdin •
Lu AA24530 •
Metitepin/Metiotepin •
Metisergid •
Pizotifen •
Ritanserin •
RS-102,221 •
S-14,671 •
SB-200,646 •
SB-206,553 •
SB-221,284 •
SB-228,357 •
SB-242,084 •
SB-243,213 •
SDZ SER-082 •
Ksilamidin
Agonisti :
Lisergamidi :
Dihidroergotamin •
Ergotamin •
Lisurid •
LSD •
Mesulergin •
Metergolin •
Metisergid ;
Triptamini :
2-Metil-5-HT •
5-BT •
5-CT •
5-MT •
Bufotenin •
E-6801 •
E-6837 •
EMD-386,088 •
EMDT •
LY-586,713 •
N -Metil-5-HT •
Triptamin ;
Drugi :
WAY-181,187 •
WAY-208,466 Antagonisti :
Antidepresanti :
Amitriptilin •
Amoksapin •
Klomipramin •
Doksepin •
Mianserin •
Nortriptilin ;
Atipični antipsihotici :
Aripiprazol •
Asenapin •
Klozapin •
Fluperlapin •
Iloperidon •
Olanzapin •
Tiospiron ;
Tipični antipsihotici :
Hlorpromazin •
Loksapin ;
Drugi :
BGC20-760 •
BVT-5182 •
BVT-74316 •
EGIS-12,233 •
GW-742,457 •
Ketanserin •
Latrepirdin •
Lu AE58054 •
Metitepin/Metiotepin •
MS-245 •
PRX-07034 •
Ritanserin •
Ro04-6790 •
Ro 63-0563 •
SB-258,585 •
SB-271,046 •
SB-357,134 •
SB-399,885 •
SB-742,457 Agonisti :
Lisergamidi :
LSD ;
Triptamini :
5-CT •
5-MT •
Bufotenin ;
Drugi :
8-OH-DPAT •
AS-19 •
Bifeprunoks •
LP-12 •
LP-44 •
RU-24,969 •
Sarizotan Antagonisti :
Lisergamidi :
2-Bromo-LSD •
Bromokriptin •
Dihidroergotamin •
Ergotamin •
Mesulergin •
Metergolin •
Metisergid ;
Antidepresanti :
Amitriptilin •
Amoksapin •
Klomipramin •
Imipramin •
Maprotilin •
Mianserin ;
Atipični antipsihotici :
Amisulprid •
Aripiprazol •
Klozapin •
Olanzapin •
Risperidon •
Sertindol •
Tiospiron •
Ziprasidon •
Zotepin ;
Tipični antipsihotici :
Hlorpromazin •
Loksapin ;
Drugi :
Butaklamol •
EGIS-12,233 •
Ketanserin •
LY-215,840 •
Metitepin/Metiotepin •
Pimozid •
Ritanserin •
SB-258,719 •
SB-258,741 •
SB-269,970 •
SB-656,104 •
SB-656,104-A •
SB-691,673 •
SLV-313 •
SLV-314 •
Spiperon •
SSR-181,507