4-MeO-DMT
Prijeđi na navigaciju
Prijeđi na pretragu
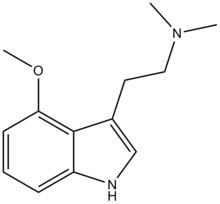
| |||
| (IUPAC) ime | |||
|---|---|---|---|
| 2-(4-metoksi-1H-indol-3-il)-N,N-dimetiletanamin | |||
| Klinički podaci | |||
| Identifikatori | |||
| CAS broj | 3965-97-7 | ||
| ATC kod | nije dodeljen | ||
| PubChem[1][2] | 12017578 | ||
| ChemSpider[3] | 23126449 | ||
| ChEMBL[4] | CHEMBL32029 | ||
| Hemijski podaci | |||
| Formula | C13H18N2O | ||
| Mol. masa | 218,29 g/mol | ||
| SMILES | eMolekuli & PubHem | ||
| |||
| Farmakoinformacioni podaci | |||
| Trudnoća | ? | ||
| Pravni status | |||
4-Metoksi-N,N-dimetiltriptamin (4-MeO-DMT), je triptaminski derivat koji deluje u životinjskim modelima na sličan način sa srodnim halucinogenim triptaminskim lekovima, mada sa znatno nižom potencijom od 5-MeO-DMT i 4-hidroksi-DMT (psilocina).[5][6][7][8] Homolog dužeg lanca 4-MeO-DET nije aktivan kod ljudi u dozama do 30 mg oralno ili pušenjem. Klinička ispitivanja su pokazala da je njegov izomer 4-MeO-MiPT aktivan u maloj meri.[9]
Reference[uredi | uredi kod]
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519.
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Hettne KM, Williams AJ, van Mulligen EM, Kleinjans J, Tkachenko V, Kors JA. (2010). „Automatic vs. manual curation of a multi-source chemical dictionary: the impact on text mining”. J Cheminform 2 (1): 3. DOI:10.1186/1758-2946-2-3. PMID 20331846.
- ↑ Gaulton A, Bellis LJ, Bento AP, Chambers J, Davies M, Hersey A, Light Y, McGlinchey S, Michalovich D, Al-Lazikani B, Overington JP. (2012). „ChEMBL: a large-scale bioactivity database for drug discovery”. Nucleic Acids Res 40 (Database issue): D1100-7. DOI:10.1093/nar/gkr777. PMID 21948594.
- ↑ Glennon RA, Young R, Benington F, Morin RD (February 1982). „Hallucinogens as discriminative stimuli: a comparison of 4-OMe DMT and 5-OMe DMT with their methythio counterparts”. Life Sciences 30 (5): 465–7. DOI:10.1016/0024-3205(82)90463-5. PMID 6801410.
- ↑ Kline TB, Benington F, Morin RD, Beaton JM (August 1982). „Structure-activity relationships in potentially hallucinogenic N,N-dialkyltryptamines substituted in the benzene moiety”. Journal of Medicinal Chemistry 25 (8): 908–13. DOI:10.1021/jm00350a005. PMID 7120280.
- ↑ Kline TB, Benington F, Morin RD, Beaton JM, Glennon RA, Domelsmith LN, Houk KN, Rozeboom MD (November 1982). „Structure-activity relationships for hallucinogenic N,N-dialkyltryptamines: photoelectron spectra and serotonin receptor affinities of methylthio and methylenedioxy derivatives”. Journal of Medicinal Chemistry 25 (11): 1381–3. DOI:10.1021/jm00353a021. PMID 6815326.
- ↑ Nichols DE, Glennon RA. Medicinal Chemistry and Structure-Activity Relationships of Hallucinogens, in Hallucinogens, Neurochemical, Behavioral, and Clinical Perspectives (Raven Press 1984), pp 95-142. ISBN 978-0-89004-990-7
- ↑ 4-MeO-MiPT Entry in TIHKAL