HZ-2
Prijeđi na navigaciju
Prijeđi na pretragu
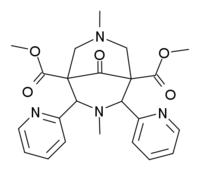
| |||
| (IUPAC) ime | |||
|---|---|---|---|
| Dimethyl 3,7-dimethyl-9-oxo-2,4-dipyridin-2-yl-3,7-diazabicyclo[3.3.1]nonane-1,5-dicarboxylate | |||
| Klinički podaci | |||
| Identifikatori | |||
| CAS broj | 253304-60-8 | ||
| ATC kod | ? | ||
| PubChem[1][2] | 356604 | ||
| Hemijski podaci | |||
| Formula | C23H26N4O5 | ||
| Mol. masa | 438.475 g/mol | ||
| SMILES | eMolekuli & PubHem | ||
| |||
| Farmakoinformacioni podaci | |||
| Trudnoća | ? | ||
| Pravni status | |||
HZ-2 je lek, koji je visoko selektivni κ-opioidni agonist.[3] On je potentan analgetik, poput morfina, sa dugotrajnim dejstvom i visokom oralnom biodostupnošću.[4][5] Nuspojave su sedacija, mučnina i disforija kao i diuretički efekti.[6]
Reference[uredi | uredi kod]
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519.
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Siener, T; Cambareri, A; Kuhl, U; Englberger, W; Haurand, M; Kögel, B; Holzgrabe, U (2000). „Synthesis and opioid receptor affinity of a series of 2, 4-diaryl-substituted 3,7-diazabicylononanones”. Journal of medicinal chemistry 43 (20): 3746–51. PMID 11020289.
- ↑ Holzgrabe, U; Cambareri, A; Kuhl, U; Siener, T; Brandt, W; Strassburger, W; Friderichs, E; Englberger, W i dr.. (2002). „Diazabicyclononanones, a potent class of kappa opioid analgesics”. Farmaco (Societa chimica italiana : 1989) 57 (7): 531–4. PMID 12164207.
- ↑ Holzgrabe, U; Brandt, W (2003). „Mechanism of action of the diazabicyclononanone-type kappa-agonists”. Journal of medicinal chemistry 46 (8): 1383–9. DOI:10.1021/jm0210360. PMID 12672238.
- ↑ Kögel, B; Christoph, T; Friderichs, E; Hennies, HH; Matthiesen, T; Schneider, J; Holzgrabe, U. (1998). „HZ2, a Selective Kappa-Opioid Agonist”. CNS Drug Reviews 4 (1): 54–70. DOI:10.1111/j.1527-3458.1998.tb00041.x.