Resveratrol
| Resveratrol | |||
|---|---|---|---|

| |||
| |||
| |||
| Drugi nazivi | trans-3,5,4'-Trihidroksistilben; 3,4',5-Stilbenetriol; trans-Resveratrol; (E)-5-(p-Hidroksistiril)resorcinol; (E)-5-(4-hidroksistiril)benzen-1,3-diol | ||
| Identifikacija | |||
| CAS registarski broj | 501-36-0 | ||
| PubChem[1][2] | 445154 | ||
| ChemSpider[3] | 392875 | ||
| UNII | Q369O8926L | ||
| DrugBank | DB02709 | ||
| KEGG[4] | |||
| ChEBI | 45713 | ||
| ChEMBL[5] | CHEMBL165 | ||
| RTECS registarski broj toksičnosti | CZ8987000 | ||
| Jmol-3D slike | Slika 1 | ||
| |||
| |||
| Svojstva | |||
| Molekulska formula | C14H12O3 | ||
| Molarna masa | 228.24 g mol−1 | ||
| Agregatno stanje | Beli prah | ||
| Tačka topljenja |
261 - 263 °C [6] | ||
| Rastvorljivost u voda | 0,03 g/L | ||
| Rastvorljivost u DMSO | 16 g/L | ||
| Rastvorljivost u etanol | 50 g/L | ||
| λmax | 304 nm (trans-resveratrol, u vodi) 286 nm (cis-resveratrol, u vodi)[7] | ||
| Opasnost | |||
| Podaci o bezbednosti prilikom rukovanja (MSDS) | Fisher Scientific[6] Sigma Aldrich[8] | ||
| R-oznake | R36 (iritatant očiju)[8] | ||
| S-oznake | S26 (u slučaju kontakta sa očima, odmah ih isprerite sa većom količinom vode i obratite se lekaru)[8] | ||
| LD50 | 23,2 µM (5,29 g)[9] | ||
|
Ukoliko nije drugačije napomenuto, podaci se odnose na standardno stanje (25 °C, 100 kPa) materijala | |||
| Infobox references | |||
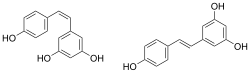
Resveratrol (3,5,4'-trihidroksi-trans-stilben) je stilbenoid, tip prirodnog fenola, i fitoaleksin koji proizvodi više biljaka kad su napadnute patogenima kao što su bakterije ili gljive.
Efekti resveratrola su tema brojnih studija na životinjama i ljudima. Njegovi efekti na životni vek mnogih model organizama su kontroverzni.[10] U eksperimentima na miševima i pacovima, zapaženo je antikancerno i antiinflamatorno dejstvo, snižavanje nivoa krvnog šećera i drugi korisni kardiovaskularni efekti. Kod ljudi, su ti efekti generalno pozitivni, ali su izraženi u manjoj meri.[11] U jednom kliničkom ispitivanju, ekstremno visoke doze (3–5 g) resveratrola, u posebno dizajniranoj formulaciji kojom se povećava biodostupnost, su znatno snizili nivoe krvnog šećera.[12] Ta 28 dana duga studija faze 1b je sprovedena u Indiji, i objavljena 2008.[13] Mada postoje trvrdnje u štampi da resveratrol manifestuje antistarosno dejstvo,[14] one za sad nisu zasnovane na rigoroznim naučnim podacima. Istraživanja resveratrola su u svojoj ranoj fazi i dugotrajni efekti suplementacije kod ljudi nisu poznati.[15][16]
Reference[uredi | uredi kod]
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519.
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Hettne KM, Williams AJ, van Mulligen EM, Kleinjans J, Tkachenko V, Kors JA. (2010). „Automatic vs. manual curation of a multi-source chemical dictionary: the impact on text mining”. J Cheminform 2 (1): 3. DOI:10.1186/1758-2946-2-3. PMID 20331846.
- ↑ Joanne Wixon, Douglas Kell (2000). „Website Review: The Kyoto Encyclopedia of Genes and Genomes — KEGG”. Yeast 17 (1): 48–55. DOI:10.1002/(SICI)1097-0061(200004)17:1<48::AID-YEA2>3.0.CO;2-H.
- ↑ Gaulton A, Bellis LJ, Bento AP, Chambers J, Davies M, Hersey A, Light Y, McGlinchey S, Michalovich D, Al-Lazikani B, Overington JP. (2012). „ChEMBL: a large-scale bioactivity database for drug discovery”. Nucleic Acids Res 40 (Database issue): D1100-7. DOI:10.1093/nar/gkr777. PMID 21948594.
- ↑ 6,0 6,1 „Resveratrol MSDS on Fisher Scientific website”. Arhivirano iz originala na datum 2012-11-03. Pristupljeno 2014-04-11.
- ↑ Camont L, Cottart CH, Rhayem Y, Nivet-Antoine V, Djelidi R, Collin F, Beaudeux JL, Bonnefont-Rousselot D (February 2009). „Simple spectrophotometric assessment of the trans-/cis-resveratrol ratio in aqueous solutions”. Anal. Chim. Acta 634 (1): 121–8. DOI:10.1016/j.aca.2008.12.003. PMID 19154820.
- ↑ 8,0 8,1 8,2 Resveratrol MSDS on www.sigmaaldrich.com
- ↑ Bechmann, LP; Zahn, D; Gieseler, RK; Fingas, CD; Marquitan, G; Jochum, C; Gerken, G; Friedman, SL i dr.. (2009). „Resveratrol amplifies profibrogenic effects of free fatty acids on human hepatic stellate cells”. Hepatology research : the official journal of the Japan Society of Hepatology 39 (6): 601–8. DOI:10.1111/j.1872-034X.2008.00485.x. PMC 2893585. PMID 19207580.
- ↑ Bass TM, Weinkove D, Houthoofd K, Gems D, Partridge L (October 2007). „Effects of resveratrol on lifespan in Drosophila melanogaster and Caenorhabditis elegans”. Mech. Ageing Dev. 128 (10): 546–52. DOI:10.1016/j.mad.2007.07.007. PMID 17875315.
- ↑ „Micronutrient Information Center: Resveratrol”. Linus Pauling Institute at Oregon State University. Pristupljeno 13. 01. 2012.
- ↑ Elliott PJ, Jirousek M (April 2008). „Sirtuins: novel targets for metabolic disease”. Curr Opin Investig Drugs 9 (4): 371–8. PMID 18393104.
- ↑ „Sirtris Announces Positive Results with Proprietary Version of Resveratrol, SRT501, in a Phase 1b Type 2 Diabetes Clinical Study - Drugs.com MedNews”. Press Release. Drugs.com. 07. 01. 2008.. Arhivirano iz originala na datum 2012-02-05. Pristupljeno 22. 02. 2012.
- ↑ „Pharma seeks genetic clues to healthy aging”. Reuters. 6. 4. 2010..
- ↑ The Connecticut Post, "Selling resveratrol: Wonder drug or snake oil?," 08/04/2009, by Melissa Healy for the Los Angeles Times news service
- ↑ Gehm BD, McAndrews JM, Chien PY, Jameson JL (December 1997). „Resveratrol, a polyphenolic compound found in grapes and wine, is an agonist for the estrogen receptor”. Proc. Natl. Acad. Sci. U.S.A. 94 (25): 14138–43. DOI:10.1073/pnas.94.25.14138. PMC 28446. PMID 9391166.
Vidi još[uredi | uredi kod]
Spoljašnje veze[uredi | uredi kod]
- Félicien Breton (2008). „Resveratrol and polyphenols in wines”. Arhivirano iz originala na datum 2013-04-14. Pristupljeno 2014-04-11.
- CTD's Resveratrol page Arhivirano 2012-05-22 na Wayback Machine-u from the Comparative Toxicogenomics Database
- U.S. National Library of Medicine: Drug Information Portal – Resveratrol
- Detailed Micro-Nutrient information on Resveratrol from the Linus Pauling Institute
- Resveratrol: Don't Buy the Hype
- Stay young on red wine drugs? Think again
- Zalet – a site that specializes in Resveratrol information
}{-}}
